AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic theory models4/3/2024  
The Rutherford atomic model does not account for the initial energy and subsequent energy level changes. The numbers of particles in an atom can be calculated from its atomic number and mass number. An atomic proposition is one that asserts that a certain thing has a certain qualitye.g., This is red. They give off electromagnetic radiation due to the circular motion of orbiting thus they must have some initial energy by the law of conservation of energy. The atomic model consists of a nucleus containing protons and neutrons, surrounded by electrons in shells. Electrons increase and decrease energy levels randomly due to the acceleration and are not always in a standard circular orbit. What is the most accurate atomic model Ans: The most accurate atomic model was given by Neil Bohr and is called Bohr’s model of atom or Planetary model.Electromagnetic radiation from the electrons in orbit would cause the atom to collapse into the nucleus in 10 -8 seconds. By the late 1800’s, John Dalton’s view of atoms as the smallest particles that made up all matter had held sway for about 100 years, but that idea was about to be challenged. 
Rutherford proposed that electrons orbit around the nucleus in set paths, but according to Maxwell’s theory, this is not possible because the atom would not be stable. This module is an updated version of Atomic Theory I. 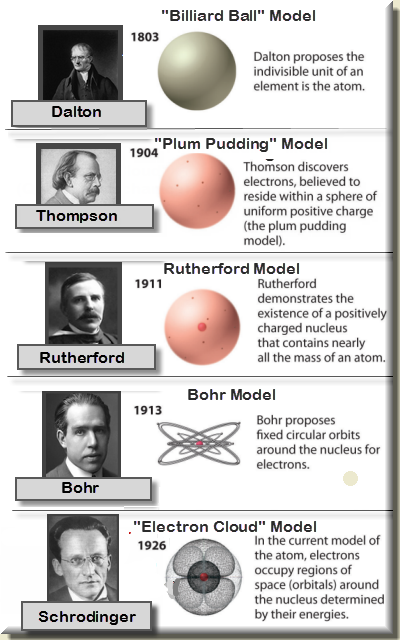
Figure A shows eight sulfur atoms, symbolized with the letter S, that are. Sulfur atoms are represented by yellow spheres. It can be represented as (a) a structural formula, (b) a ball-and-stick model, and (c) a space-filling model.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
